PSP CAPSULES IN CLINICAL CANCER THERAPY---------A BRIEF SUMMARY OF CLINICAL PHASE I, II AND III TRIALS
Polysaccharide-peptide (PSP) is a protein bound polysaccharide isolated from the COV-1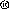 strain of Yunzhi (Coriolous versicolor mushroom) and made from modern alcohol extraction techniques. Each capsule contains 0.34 grams of PSP. Experimental in-vitro and in-vivo studies have shown PSP inhibits the proliferation of cancer cells including P338 leukemia cells, S 180 cells, Ehrlich ascites, and stomach and lung cancer cells. It also inhibits the growth of some tumors such as the lymphatic tumor of human skin tissue cells. In addition, PSP affects the immune system of mice by stimulating the production of α\interferons, increasing the phagocytic index and metabolic rate of the reticuloendothilial system and by raising the HC 50 (median hemolytic dose), IgG and PFC (plaque forming cell) values. Human in-vivo experiments have also shown PSP can modulate the immune system by helping to prevent and partly eliminate the side effects of radiation and chemotherapeutic agents used by cancer patients. strain of Yunzhi (Coriolous versicolor mushroom) and made from modern alcohol extraction techniques. Each capsule contains 0.34 grams of PSP. Experimental in-vitro and in-vivo studies have shown PSP inhibits the proliferation of cancer cells including P338 leukemia cells, S 180 cells, Ehrlich ascites, and stomach and lung cancer cells. It also inhibits the growth of some tumors such as the lymphatic tumor of human skin tissue cells. In addition, PSP affects the immune system of mice by stimulating the production of α\interferons, increasing the phagocytic index and metabolic rate of the reticuloendothilial system and by raising the HC 50 (median hemolytic dose), IgG and PFC (plaque forming cell) values. Human in-vivo experiments have also shown PSP can modulate the immune system by helping to prevent and partly eliminate the side effects of radiation and chemotherapeutic agents used by cancer patients.
Phase I Clinical Trial
In 1991, a clinical phase I study was carried out by Dr. Guo-ming Xu of Changhai Hospital in Shanghai to assess the feasibility of using this agent clinically by observing healthy human subjects taking PSP for any possible toxic side effects and adverse reactions. Twenty-one people, including 16 healthy volunteers and 5 post-operative breast cancer patients without recurrence or metastases were equally divided into three groups to take a total of two, four or six grams of PSP each day respectively. Each dosage was divided equally into capsules that were taken after each meal three times a day for 30 days. Symptoms of abdominal pain, diarrhea, constipation, poor appetite, headache, dizziness, skin rash, itching, palpitation, stuffy chest, and urinary frequency were monitored. An electrocardiogram (ECG) and laboratory parameters including peripheral blood counts (hemoglobin, white blood cells, red blood cells and platelets), liver function and renal function tests were compared before and after PSP was taken for the study duration.
Results of the phase I trial found no significant toxic side effects or adverse reactions related to PSP intake. No abnormal changes in the ECG and laboratory tests mentioned above were observed in any group taking PSP. Four people experienced a slight degree of loose bowels, which occurred less than twice a day (2 people taking 2gms of PSP/day, 1 person taking 4gms of PSP/day and 1 person taking 6gms/PSP day). Positive effects of taking PSP were also noted. Eleven people (52.8%) developed an increase of appetite, which was most noticeable in the group taking six grams of PSP/day (n=6). Based on the results of this study, PSP was considered safe to use clinically and a Phase II clinical trial was undertaken in cancer patients.
Phase II Clinical Trial
In November 1991, in accordance with the approval document No. (91)ZL-15 of the Ministry of Public Health, the Shanghai Public Health Bureau appointed the following 8 hospitals to carry out a multi-center phase II clinical trial using PSP. They were the Long-Hua Hospital, Shu-Guang Hospital of the University of Chinese Medicine, Cancer Hospital of Shanghai Medical University, Shanghai Chest Hospital, Ren-Jin Hospital of the Second Medical University of Shanghai, The Third Textile Hospital of Shanghai, the Chang-Hai Hospital and the Chang-Zheng Hospital of the Second Military Medical University. This prospective double-blind study took place from February to July 1992. The purpose was to investigate the safety and efficacy of using PSP as an adjunct to chemo and radiation therapy. Stomach, primary lung and esophagus cancers are the most prevalent malignancies found in Shanghai. Patients with these types of cancer were selected for the study. Diagnoses were confirmed by surgery, clinical tests and cell pathology reports. Patients were also diagnosed by Chinese medicine practitioners and were classified according to deficiency of vital energy, deficiency of body fluid and deficiency of heart and spleen.
Four hundred and eighty five patients were admitted to the study. Of those, 274 patients were randomly assigned to a control group (n=135) and treatment group (n=139) according to the types of cancers they had. The remaining 211 cases were admitted into an open non-random group in which the physicians knew they were being given PSP. See Table 1 for a breakdown of the types of cancer of each group. All patients received two courses of chemotherapy or radiation depending on the type of cancer. Each course lasted approximately one month. See Table 2 for treatment regimens. Patients in the treatment group received three 0.340 gram capsules of PSP three times a day. Patients in the open group also received the same PSP dosing schedule as patients in the treatment group. The control group was given three 50mg capsules of Batilol in the same packaging as the PSP three times a day. Therapy was initiated in all groups when chemotherapy or radiation was started and lasted for approximately two months.
Table 1
| Cancer Type |
Pathology |
Total No. of Patients |
Treatment group |
Control Group |
PSP open group |
| Esophagus |
Squamous cell carcinoma |
172 |
56 |
56 |
60 |
| Stomach |
Adenocarcinoma |
149 |
36 |
34 |
79 |
| Undifferentiated squamous cell |
13 |
3 |
5 |
5 |
| Lung |
Squamous cell |
73 |
20 |
23 |
30 |
| Adenocarcinoma |
78 |
20 |
21 |
37 |
| Total |
|
485 |
135 |
139 |
211 |
Table 2
| Cancer Type |
Drug |
Dose |
Route |
Frequency |
#of cycles |
Stomach Cancer
(MF Plan) |
Mitomycin C |
6-8mg |
IV |
Day 1 of 1month cycle |
2 |
| 5FU |
500-750mg |
Ivgtt |
Days 1-5 of 1 month cycle |
- |
| Lung Cancer(MAP Plan) |
Mitomycin C |
10mg |
IV |
Day 1 of 1month cycle |
2 |
| ---- |
Adriamycin |
40mg/m2 |
Ivgtt |
Day 1 of 1 month cycle |
- |
| Metaclopromide |
20 mg |
Ivgtt |
Day 1 of 1 month cycle |
- |
| DDP |
60-90mg/m2 |
Ivgtt |
Day 1 of 1 month cycle |
- |
| Vincristine (used instead of Adriamycin if patient had heart disease) |
1mg/m2 |
IV |
Day 1 of 1 month cycle |
- |
| Esophagus Cancer |
Radiotherapy8 Mv X-ray or
Co60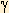 -ray -ray |
65-70 GY |
- |
6-7 weeks |
- |
Clinical symptoms of weakness, feeling tired, poor appetite, nausea and vomiting, mouth and throat dryness, anxiety, insomnia, insomnia, palpitations, pain, shortness of breath, spontaneous or night sweating, tongue appearance and pulse quality were evaluated monthly. Body weight and Karnofsky's performance standard (a quality of life assessment tool) were evaluated before and after treatment. Hematological parameters such as WBC's (white blood cells), Hgb (hemoglobin), platelets, liver and renal function tests and immuno assays such as NK (natural killer) cells, interleukin 2 (IL-2), T cells and their subgroups with the exception of the WBC's were all measured before and after treatment as well as on a monthly basis. Clinical symptoms and laboratory results were validated by the Chairman of TCM Clinical Research under the China's Department of Health for both the phase II and phase III clinical trials.
Treatment with PSP was considered "effective" if clinical symptoms were markedly improved and the blood and immunological indexes remained stable or improved by a third or more. It was also considered "effective" if clinical symptoms markedly improved and the Karnofsky score or body weight remained equal or was improved compared to the control group. Treatment that had a "marked effect" had to satisfy all of the above criteria. Study results were recorded and analyzed according to the Bonferrori x2(Gtest), qualitative x2, and Ridit or Mann Whitney Wilcoxon U tests. Statistical significance was set at p<0.05.
The overall response rate (combination of patients which had treatment that was "effective" or "markedly effective") was significantly higher in the treatment group (82.0%) and open group (83.9%) compared to the control group (45.2%), (p< 0.001). When each group was further analyzed by the types of cancer, the PSP treated and open group still responded better then the control group regardless of the type of cancer (p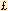 G0.01). See Table 3 for results. G0.01). See Table 3 for results.
Overall Effectiveness of Treatment |
| |
Stomach Cancer |
Lung Cancer |
Esophagus Cancer |
| Control |
32.14% |
32.50% |
76.92% |
| Treatment |
80.36% |
70.45% |
97.44% |
| Open |
78.33% |
71.64% |
97.62% |
No statistically significant differences were found between the treatment, open and control groups in terms of changes in WBC's, Hgb, and platelets. The majority of the three groups had a stable hematological profile at the end of treatment period showing PSP had no adverse effect on these parameters in patients undergoing chemotherapy and radiation treatment. An important finding was the effectiveness of PSP in improving the immunological profiles of the patients. The rate of increase in activity of natural killer cells (NK) in the treatment (63.97%) and open groups (68.93%) was significantly higher than the control group (4.11%) (p<0.001). The treatment group also had a significant increase in the amount of interleukin 2 (IL-2) produced after taking PSP (p<0.001), whereas the control group did not (p>0.05). In addition, there was a tendency for the CD+4/CD+8 ratio to improve or remain stable in more PSP treated patients then control patients. No obvious renal, hepatic or cardiac toxicities were seen during the study period. Overall, the Phase II clinical trial showed PSP is safe and can help lessen the degree of side effects that chemotherapy and radiation produces. It also improved or maintained the immunological profiles of stomach, esophagus and lung cancer patients.
Phase III Clinical Trial
Based on the PSP's significant findings in the investigated cancers of the Phase II trial, permission was granted by the Chinese Administration of Health Bureau to carry out a multi-center Phase III clinical trial. Fourteen hospitals including the eight who participated in the phase II trial conducted this randomized study from April 1996 to September 1997.
Six hundred and fifty patients with either stomach, lung or esophageal cancer entered the study. Standard cancer treatment mainly consisted of surgery, chemotherapy and/or radiation therapy. Chemotherapy and radiation treatment lasted for approximately two months and followed the same regimens as the phase II trial. See Table 2. Patients were randomly divided into a treatment (n=96) and control group (n=93). The open group consisted of 461 patients where both the physicians and patients knew they were taking PSP. See Table 4 for a breakdown of each type of cancer. Patients in the treatment and open group received one gram of PSP three times a day for two months. The control group received 150mg Batilol three times a day for two months. Both therapies were given concurrently with the start of chemotherapy or radiation therapy. Evaluation of effectiveness of treatment was the same as the phase II trial.
Table 4
| Type of cancer |
No. Control Group |
No. Treatment Group |
No. Open Group |
| Stomach |
30 |
30 |
170 |
| Lung |
33 |
35 |
129 |
| Esophagus |
30 |
31 |
162 |
| Total |
93 |
96 |
461 |
The Phase III trial showed similar results to the Phase II trial. The overall response rate (combination of patients which had treatment that was "effective" or "markedly effective") for the treatment group (87.5%) was significantly higher than the control group (41.9%) (p<0.01). The open group also had an 85.5% overall response rate. Like the phase II, the overall response of treatment group patients with stomach, lung and esophagus cancer was also significantly higher when compared with the same cancers of the control group (p<0.01). See Table 5 for overall effectiveness of PSP in the different types of cancer.
Table 5
Overall Effectiveness of Treatment |
| |
Stomach Cancer |
Lung Cancer |
Esophagus Cancer |
| Control Group |
42.4% |
42.4% |
43.3% |
| Treatment Group |
90% |
85.7% |
87.0% |
| Open Group |
85.3% |
86.0% |
85.2% |
The treatment group experienced a highly significant decrease (p<0.01) in symptoms of fatigue, loss of appetite, and mouth or throat dryness and a significant decrease in anorexia and vomiting, sweating, and pain (p<0.05) compared to the control group. The Karnofsky performance status is used as an evaluation of quality of life. The majority of patients taking PSP, n=79 in the treatment group, n=257 in the open group and n=65 in the control group had a stable Karnofsky score (score change of less than 10) after treatment. However, the control group had a 22.6% decrease in Karnofsky scores after treatment compared with only 5.2% in the treatment group and 3.7% in the open group. This result suggests PSP helps cancer patients to maintain their quality of life and level of functioning after chemotherapy and radiation. A weight increase of one kilogram or more was seen in 40% of the treatment group and 47.2% in the open group as compared to only 15.1 % in the control group (p<0.05).
While there were decreases in the WBC and Hgb levels after chemotherapy and radiation in all the groups tested, the decreases were significantly less in the treatment group compared with the control group (p<0.01). No significant changes were seen in the RBC (red blood cell) levels. The CD4+/CD8+ ratios remained stable after taking PSP in both the treatment and open groups. However, the control group had a significantly lower CD4+/CD8+ ratio (1.16 0.59) compared to the treatment group (1.61 0.59) compared to the treatment group (1.61 0.77) after taking PSP (p<0.01). Unlike the phase II trial, no significant changes in NK activity were seen in any of the tested patients before or after treatment. Both the treatment and open group experienced an increase in IL-2 production after treatment with PSP. The increase in the treatment group went from 32.84 0.77) after taking PSP (p<0.01). Unlike the phase II trial, no significant changes in NK activity were seen in any of the tested patients before or after treatment. Both the treatment and open group experienced an increase in IL-2 production after treatment with PSP. The increase in the treatment group went from 32.84 16.39 to 37.59 16.39 to 37.59 19.73 (p<0.05), which was significantly higher then the control group (31.94 19.73 (p<0.05), which was significantly higher then the control group (31.94 12.65) after taking Batilol (p<0.01). Among the 650 patients tested in the trial, there were no significant changes seen the heart, liver and renal function after treatment with either PSP or Batilol from their baseline functioning. 12.65) after taking Batilol (p<0.01). Among the 650 patients tested in the trial, there were no significant changes seen the heart, liver and renal function after treatment with either PSP or Batilol from their baseline functioning.
The results of this phase III clinical trial were mostly consistent with the phase II clinical trial results. PSP was shown to be helpful in easing the side effects associated with chemotherapy and radiation. Biologically, PSP exhibited a protective effect on maintaining the immunological functions of patients receiving immunosuppressive radiation and chemotherapy. Thus, PSP can be classified as a clinical biological response modifier. The phase III clinical also demonstrated PSP can be used safely among stomach, lung, and esophagus cancer patients with no adverse effects on heart, renal or liver functions.
Conclusion
Besides the patients studied in the prospective clinical trials, individuals with other malignancies such as nasopharyngeal carcinoma, melanoma, colon rectal cancer, cervical cancer, lymphoma, hepatoma, breast cancer and others have used PSP capsules during or after surgery, chemotherapy and radiation therapy in China and Hong Kong for several years. Most of these individuals report feeling improvement in their general condition, appetite, energy level, and ability to digest food. As a biological response modifier, PSP may help them to improve or maintain their immune status while decreasing the severity of the side effects associated with chemotherapy and radiation. Its safety profile also makes it an ideal adjunct therapy to help in the treatment of cancer. Further research is necessary to verify these findings in other malignancies.
Written By:
Professor Qing-yao Yang
Head, Institute of Microbiology and Immunology
Shanghai teachers University
References:
| 1. |
Yang, Q.Y. & Kwok C.Y. (Eds.) 1993 PSP International Symposium, Shanghai: Fudan University Press (1993). |
| 2. |
Yang, Qing-yao (Ed.) Advanced Research in PSP, Hong Kong: The Hong Kong Association for Health Care (1999). |
For print version
|